SAMPLE CAPSULES
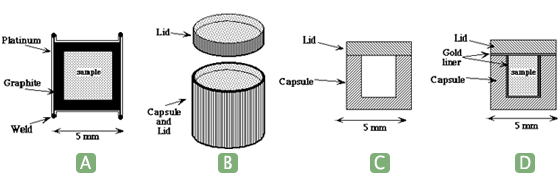
The choice of sample container is crucial to a successful synthesis. It should be chemically inert, and provide a barrier to exchange of material between the sample and its surroundings. Containers can be sealed or unsealed, depending on the sample. For instance, for oxide ceramics not subject to redox reactions and containing low vapor pressure components, a sample container is not needed so the graphite furnace can be used with graphite disks placed at each end of the sample. This configuration maximizes the sample volume. Alternatively, graphite or boron nitride crucibles and lids can be used, but these are permeable to fluids, so volatiles such as CO2 or N2 will be at least partially lost from the sample. Sealed containers prevent gain or loss of all volatiles except hydrogen.
The most common sealed capsule is made of thin platinum, gold, or palladium-silver alloy tubing with end caps of the same material welded together using an electric arc (Fig. 1a and 1b). The compatibility of the metal capsule with the sample must be carefully considered. Many transition metals are very soluble in platinum, causing their oxides to disproportionate, for example ferrous iron dissolves in platinum by the reaction 3 FeO = Fe(Pt) + Fe2O3. Reactions like this change both the bulk composition and oxidation state of the sample.
Sealed containers can also be fabricated into thick-walled crucibles with flat lids (Fig. 1c). This works well when it is desired to saturate the system in a metal. For instance Ti metal capsules were used to investigate a portion of the system Ti – C – Si by Sambasivan and Petuskey (1992). A variation is to line the capsule with platinum or gold (Fig. 1d). Alternatively, thick-walled silver capsules can be fabricated from rod stock.
Hydrogen is a nemesis in all high-pressure experiments because of its very high permeability in metals, the difficulty of removing all water from furnace assemblies, and the reaction between water and the graphite furnace to produce hydrogen. Hydrogen will diffuse either in or out of the sample container depending on the gradient in hydrogen chemical potential. This can be used to advantage to partially dehydrogenate samples by placing a hydrogen sink such as Fe2O3 outside the sample container. It can also be a serious disadvantage if hydrogen diffuses into the capsule and reduces cations in high oxidation states. Oxygen activity can be controlled by, for example, the coexistence of a metal and its oxide. For instance, silver metal together with Ag2O fixes oxygen activity at very high values. Other techniques to fix oxygen activity in high pressure runs, as well a detailed discussion of all sample capsule chemistry and configurations can be found in Simulating the Earth by Holloway and Wood (1988).
